 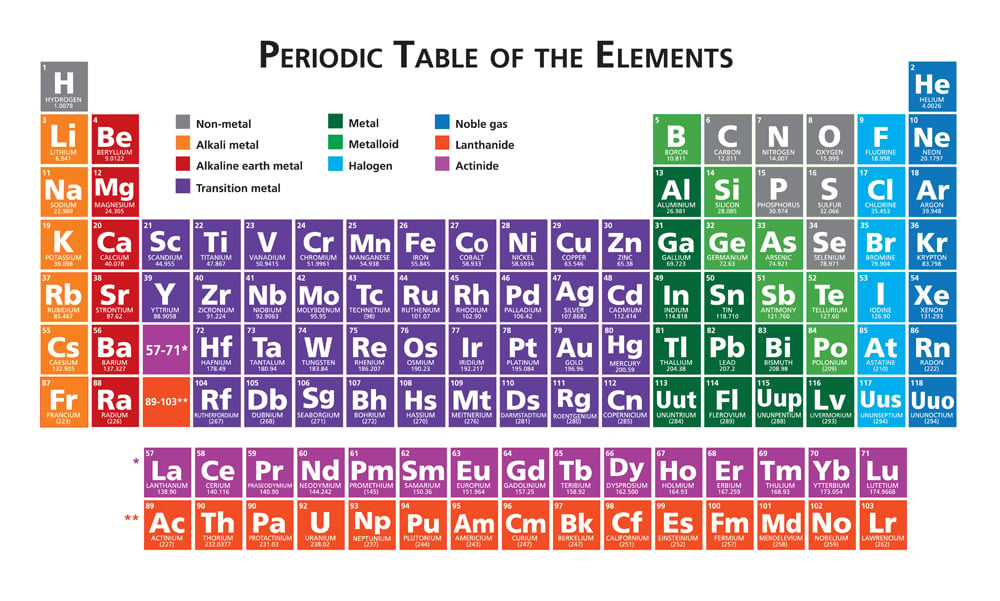
Define the Periodic Table with elements name.Atomic number of a few elements are given below 10, 20, 7, 14(a) Identify the elements(b) Identify the Group number of these elements in the Periodic Table(c) Identify the Periods of these elements in the Periodic Table(d) What would be the electronic configuration for each of these elements?(e) Determine the valency of these elements.(a) What is the usual number of valence electrons and valency of group 18 elements of the periodic table?(b) What happens to the number of valence electrons in the atoms of elements as we go down in a group of the periodic table?.(a) How does the electropositive character of elements change on going down in a group of the periodic table?(b) State how the valency of elements varies (i) in a group, and (ii) in a period, of the periodic table.In the modern periodic table, which are the metals among the first ten elements?.Compare and contrast the arrangement of elements in Mendeleev’s Periodic Table and the Modern Periodic Table.What is the major characteristic of the first elements in the periods of the periodic table? What is the general name of such elements?.Name of 118 elements in the periodic table.How does the valency of elements change on moving from left to right in the third period of the periodic table?.How does the valency of elements vary in going down a group of the periodic table?.Valence electrons: The electrons in the highest occupied principal energy level of an atom. Inner-shell electrons: Those electrons that are not in the outer shell and are not involved in the reactivity of the element. The periods (rows) of the periodic table indicate the number of shells that surround a nucleus.The group numbers (columns) of the periodic table indicate the total number of outer electrons in the valence shell.Valence electrons govern many chemical properties, reactivity, and bonding.Valence electrons are the outermost electron in an electron configuration.These are named after the orbitals, so there is an s-block, p-block, d-block and f-block.Ĭhemistry Question Pack Passage 19 Question 106 The periodic table can be broken up into different blocks based on which orbitals their valence electrons occupy. 
Thus, group number is a good predictor of how reactive each element will be. Group 1 elements have just one valence electron while group 18 elements have eight (except for helium which has only two electrons total). In general, the number of valence electrons is the same within a column and increases from left to right within a row. 
Similarly, an element’s column number gives information about its number of valence electrons and reactivity. In addition, the position of an element in the periodic table-its column, or group, and row, or period-provides useful information about how those electrons are arranged.Ĭonsidering the first three rows of the table, each row corresponds to the filling of a different electron shell: helium and hydrogen place their electrons in the 1n shell, while second-row elements like Li start filling the 2n shell, and third-row elements like Na continue with the 3n shell. In a neutral atom, the number of electrons will equal the number of protons. 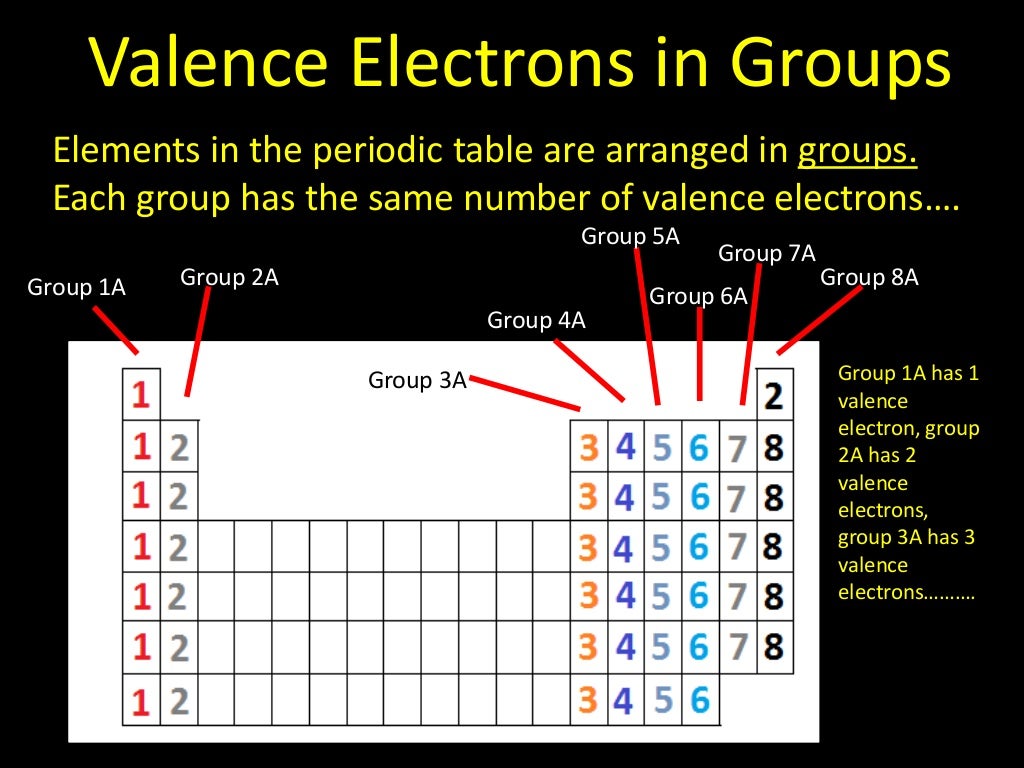
Remember, elements are placed in order on the periodic table based on their atomic number. Inner-shell electrons are not involved directly in the element’s reactivity or in the formation of compounds. In general, atoms are most stable and least reactive when their outermost electron shell is full. Valence electrons, the electrons in the outermost or valence shell, are important as they provide insight into an element’s chemical properties and are the ones gained, lost, or shared during a chemical reaction. The properties of an atom relate directly to the number of electrons in various orbitals. Valence electrons are the electrons in the highest occupied principal energy level of an atom.Įlements are organized by period and group, with the period corresponding to the principal energy level and the group relating to the extent the subshells are filled. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
